A commonly available oral diuretic pill approved by the Food and Drug Administration may be a potential candidate for people with a genetic risk for Alzheimer’s disease.
Dr. Nestor Galvez, a neurologist with the Cleveland Clinic Weston, said the research included prior studies on animals along with the use of artificial intelligence.
The combination helped pinpoint which particular diuretic drug currently being prescribed to patients for other conditions may also be beneficial for Alzheimer’s.
“What they found is this particular drug, bumetanide, seems to prevent the pathological changes you see in people with Alzheimer’s, but you have to have a specific genotype, the APOE, in order to prove that,” Galvez said.
- An FDA-approved drug used for high blood pressure and swelling, called bumetanide, alleviated symptoms of Alzheimer’s disease in mice.
- Electronic health records revealed a lower prevalence of Alzheimer’s disease among people who’d taken the drug.
- The finding suggests that bumetanide warrants further investigation as a possible treatment for Alzheimer’s disease.
Alzheimer’s disease (AD), the leading cause of dementia, has no effective therapies. People with AD differ in their genetic makeup, pathology, and clinical symptoms. This makes traditional drug development for AD difficult. Developing approaches for distinct subpopulations of patients might be a more productive approach.
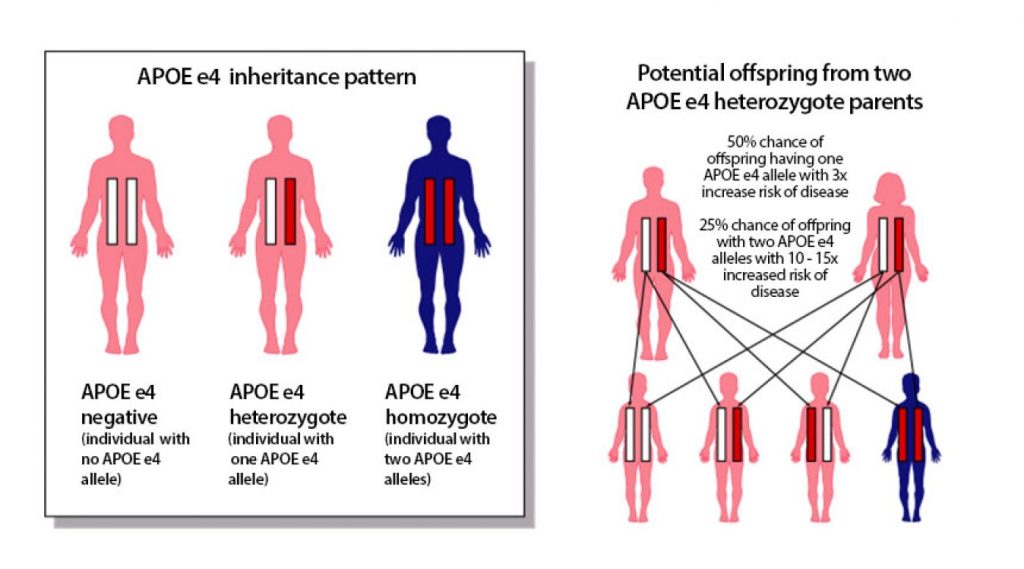
The greatest genetic risk factor for late-onset AD occurs in a gene called APOE. Having one copy of a particular form of this gene, called APOE4, increases AD risk by three- to four-fold. Having two copies increases the risk as much as 14-fold.

A team of researchers led by Drs. Marina Sirota from the University of California, San Francisco, and Yadong Huang from the Gladstone Institutes sought to find a drug that could help patients with AD carrying APOE4. NIH’s National Institute on Aging (NIA) primarily supported the work. Results appeared in Nature Aging on October 11, 2021.
The team began by comparing gene expression between brain samples from patients with AD and non-AD controls. They identified a set of changes that were unique to people with APOE4. Next, the team looked for existing drugs that might reverse these expression changes. To do so, they consulted a database with information on how more than 1,300 existing drugs affect gene expression.
The FDA-approved drug bumetanide stood out as the best candidate for further investigation. Bumetanide is used to treat high blood pressure and swelling caused by heart, liver, or kidney disease. It has also been investigated for brain disorders such as seizures, autism, depression, and schizophrenia. This suggests that it might have effects on the central nervous system that could impact AD.
The researchers tested the effects of bumetanide on mouse models of AD with APOE4. These mice have abnormal electrical activity in neurons of the hippocampus, the brain region associated with memory formation. They also have impaired spatial learning. Bumetanide treatment restored normal neuronal activity and reduced spatial learning problems.
Beta-amyloid plaques in the brain are one of the hallmarks of AD. In mice with plaques, bumetanide treatment significantly reduced the number of plaques.
The researchers confirmed that bumetanide reversed AD-associated gene expression changes in the mice. Bumetanide had a similar effect on gene expression in cultured APOE4 human neurons.
Finally, the researchers used electronic health records to compare AD prevalence and orders for bumetanide among people over the age of 65. They found a 35-75% lower prevalence of AD in people who were exposed to bumetanide compared with those who weren’t.
The results suggest that bumetanide warrants further investigation as an AD treatment. Controlled clinical trials will be needed to confirm the drug’s efficacy in humans.
“Our study highlights the power of combining computational drug repurposing with precision medicine,” Huang says. “This may be a really efficient way to find better drugs for Alzheimer’s and other complex age-related diseases.”

